JANUARY 2022 INTAKE – AUSTRALIA
We are internationally recognized as a leading regional full-service contract research organization (CRO) in Asia-Pacific providing clinical development services across all clinical trial phases and a broad range of therapeutic areas. We have created a strong benchmark, in the healthcare industry. We are a biotech focused CRO. with strong experience in a broad range of therapeutic areas including:
- Oncology
- Immunology
- Infectious Diseases
- Vaccines
- Orphan and Rare Diseases
- Neurology and Psychiatry
- Cardiovascular
- Metabolic
- Endocrinology
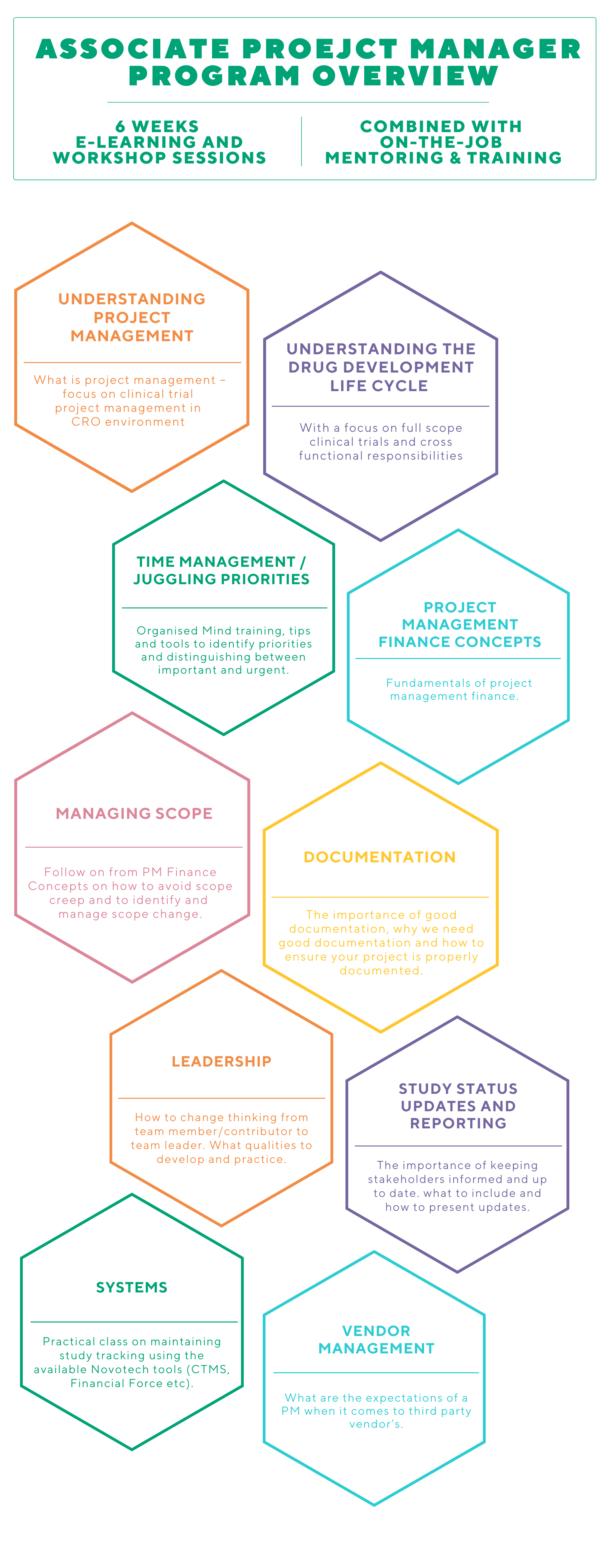
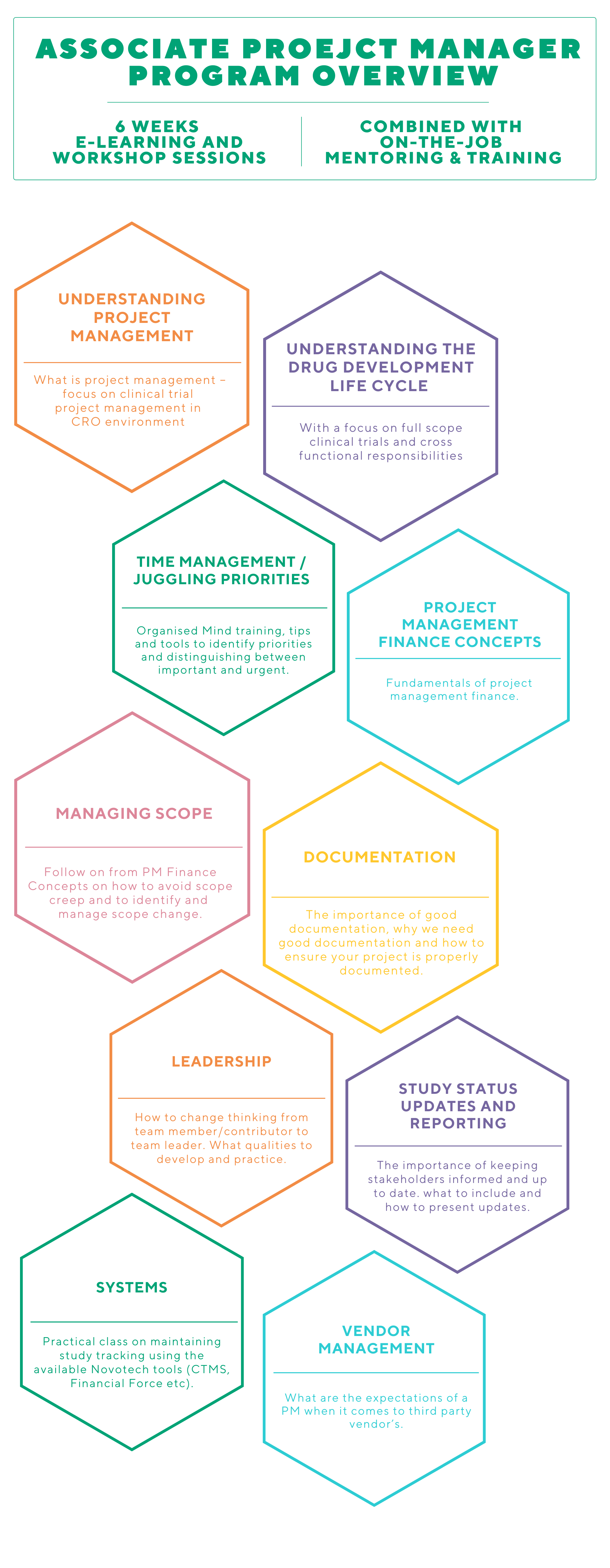
WHAT IS THE aPM PROGRAM?
The Associate Project Manager (aPM) program is a targeted training program that connects LCRAs and Project Specialists to the Project Manager career pathway. The program is delivered in a group training format to enable participants to support each other and build a network for ongoing support. The APM program will support your career progression with the following experience in clinical trials:
- Experience in direct clinical trials working in various study phases (I, II, III) and stages (e.g. start-up, maintenance and close-out) in CRO, pharma, biotech or research.
- Experience as a Project Coordinator, Study Start Up or Project Specialist with monitoring experience highly regarded.
- Competency in standard business procedures (SOPs, global regulations, CTMS). Previous matrix management and CRO experience advantageous.
Training is delivered over 6 weeks with a mix of Virtual Live Sessions / Workshops, self-paced learning and on the job experience. Each week there is a live workshop Check-In Session with an open forum where you can share your experiences, ask questions, request additional learning, seek clarification etc.
You will be paired with an experienced Novotech Project Manager as your NovoBuddy to provide guidance that supplements the program and management support and give you a “go to person” to help deal with day-to-day questions as they arise.
Apply Now
Apply Now
WHAT TO EXPECT AS AN APM
As an APM, you will be responsible for all aspects of study conduct in assigned countries/regions within larger multi-country projects and you will act as a link between the PM and the local study teams, including LCRAs (where assigned) and CRAs. You will support the Project Manager in the planning, management and execution of project management responsibilities including client relationship management.
The role is broad and varied. Ideally, you will have exposure to a range of trials at various phases and stages with experience managing across Asia. As a Novotech APM, you will have a key role in shaping our business and reputation as a highly regarded CRO. You will be part of an expanding team, due to our continued success in securing new studies.
NOVOTECH’S COMMITMENT TO CAREER PROGRESSION
Team members at Novotech have ownership of their role and are trusted to deliver. Through ongoing learning, training, development, and structured career pathways, you will be supported to continue growing in your career.
There will be ample opportunity for you to progress your career and learn from some of the most experienced PMs in the industry. Our clinical professionals are immensely talented and well regarded in their field and you will be working alongside these individuals on cutting-edge therapeutic solutions.
Apply Now